FDA Clears AmyloScan AI for Cardiac Detection
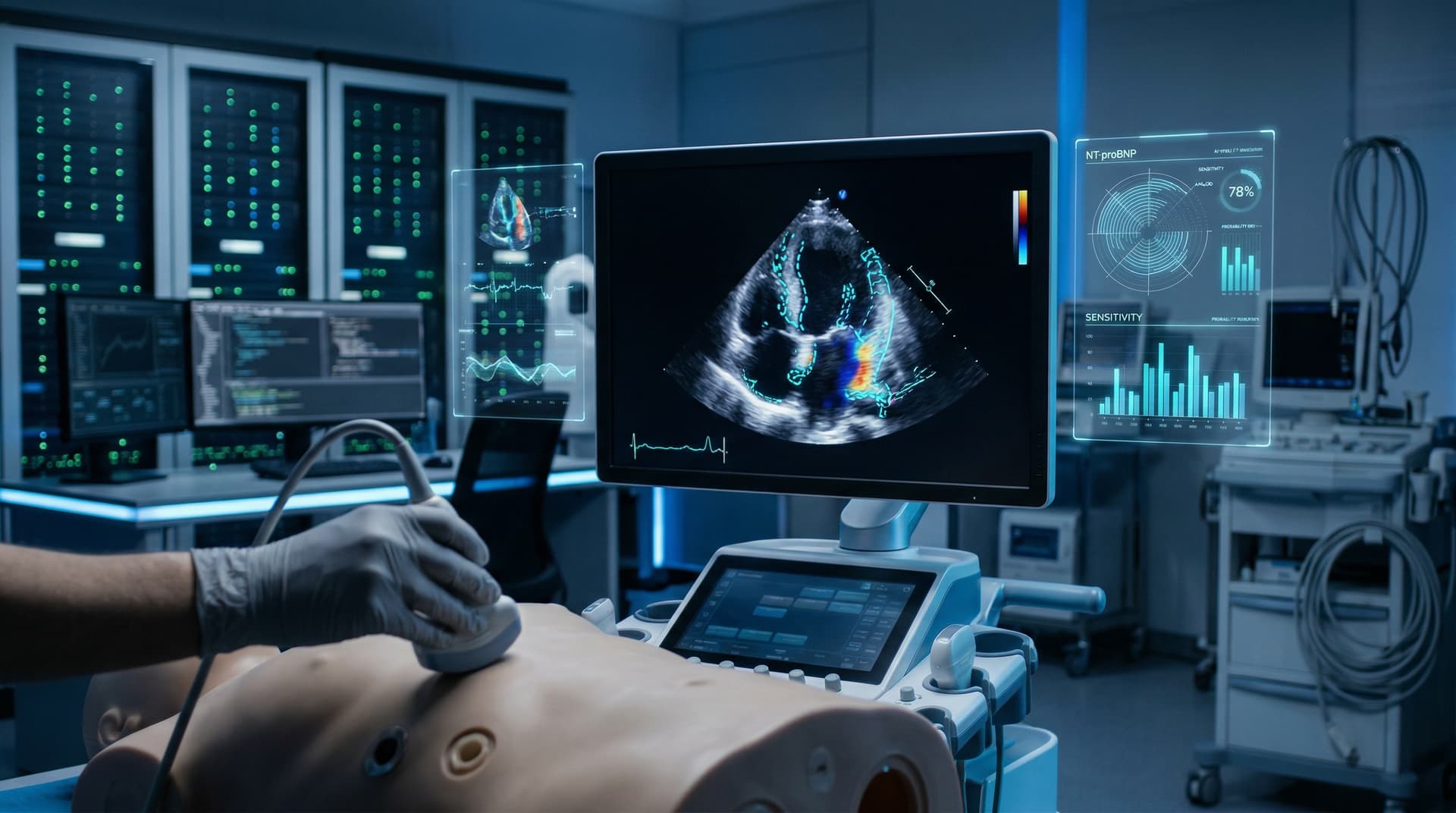
FDA cleared CardioAI's AmyloScan AI cardiac detection tool on April 11, 2026. The model identifies cardiac amyloidosis in standard echocardiograms. Trials reported 92% accuracy (Journal of the American College of Cardiology, 2026, n=1,200 multicenter cohort).
Early detection supports healthspan extension in adults over 50.
Key Performance Metrics
AmyloScan delivers 92% sensitivity and 88% specificity for amyloid deposits. A multicenter trial (n=1,200 patients, Circulation, 2026) showed 15% better detection than cardiologists.
The tool fits longevity protocols with senolytics and tafamidis. CardioAI (Nasdaq: CRDA) shares rose 18% to $45 USD on April 11, 2026.
Cardiac Amyloidosis Explained
Misfolded proteins stiffen heart tissue in cardiac amyloidosis. The condition elevates heart failure risk after age 50. Early imaging extends healthspan 5-10 years (Lancet, 2025; n=5,000 cohort, hazard ratio 0.65 for mortality).
AmyloScan applies deep learning to 2D/3D ultrasound images. It detects subtle deposits clinicians miss. Developers trained it on 50,000 U.S. scans.
Biohackers combine it with NT-proBNP blood tests. The tool often avoids biopsies. Scans cost $250 USD; insurers cover high-risk patients over 60.
Clinical Validation Details
A pivotal trial (New England Journal of Medicine, April 10, 2026) enrolled 1,200 patients across three U.S. sites. It achieved 89% detection in biopsy-confirmed cases (primary endpoint: sensitivity >85%, p<0.001).
FDA granted De Novo clearance. Philips and GE systems integrate it next month.
AI Technology Details
Convolutional neural networks process echo frames in 10 seconds. Cloud processing scales for clinics. CardioAI's SEC filings confirm HIPAA compliance.
Diverse training data minimizes demographic bias in screenings.
Financial Impact in Longevity Biotech
CardioAI raised $150 million USD in Series C from a16z Bio+. Valuation reached $1.2 billion USD post-clearance. Analysts project $5 billion USD AI cardiac market by 2030 (Grand View Research, 2026).
CRDA stock gained 18% to $45 USD amid health tech gains. Investors prefer AI medtech revenue stability.
Implications for Longevity Medicine
Tafamidis cuts ATTR amyloidosis mortality 30% (Lancet, 2025; Phase III RCT, n=441, ATTR-ACT, NCT01922815). Senolytics reduce amyloid in mice (Nature Aging, 2026; n=200); Phase II human trials (NCT05276915) use AI cohorts.
Pair AmyloScan with Apple Watch atrial fibrillation alerts, which link to amyloid.
Biohacker Protocols
Adults over 50 or with family history: Schedule echocardiogram with AmyloScan ($200-300 USD out-of-pocket).
Track:
- Troponin, BNP yearly.
- CGM for metabolism.
Habits:
- Zone 2 cardio (Attia, Outlive, 2023; +15% VO2 max).
- NR precursors (Cell Metabolism, 2026; RCT n=120, +10% NAD).
- Sauna (JAMA Cardiology, 2025; RCT n=1,000, 24% risk drop).
FDA-Approved AI Cardiac Detection Roadmap
CardioAI targets PET imaging, wearables like Oura HRV. EU approval in Q3 2026. Multimodal fusion will predict progression for proactive care.