- 1. Al-Jabbar et al. Cureus 2026 review (n=5,000+ scans) hits 95% AI sensitivity for lung nodules.
- 2. BTC reaches $74,628 (+0.2%), Fear & Greed at 23 (extreme fear), April 15, 2026.
- 3. Biohackers combine AI LDCT with hs-CRP and NMN for healthspan.
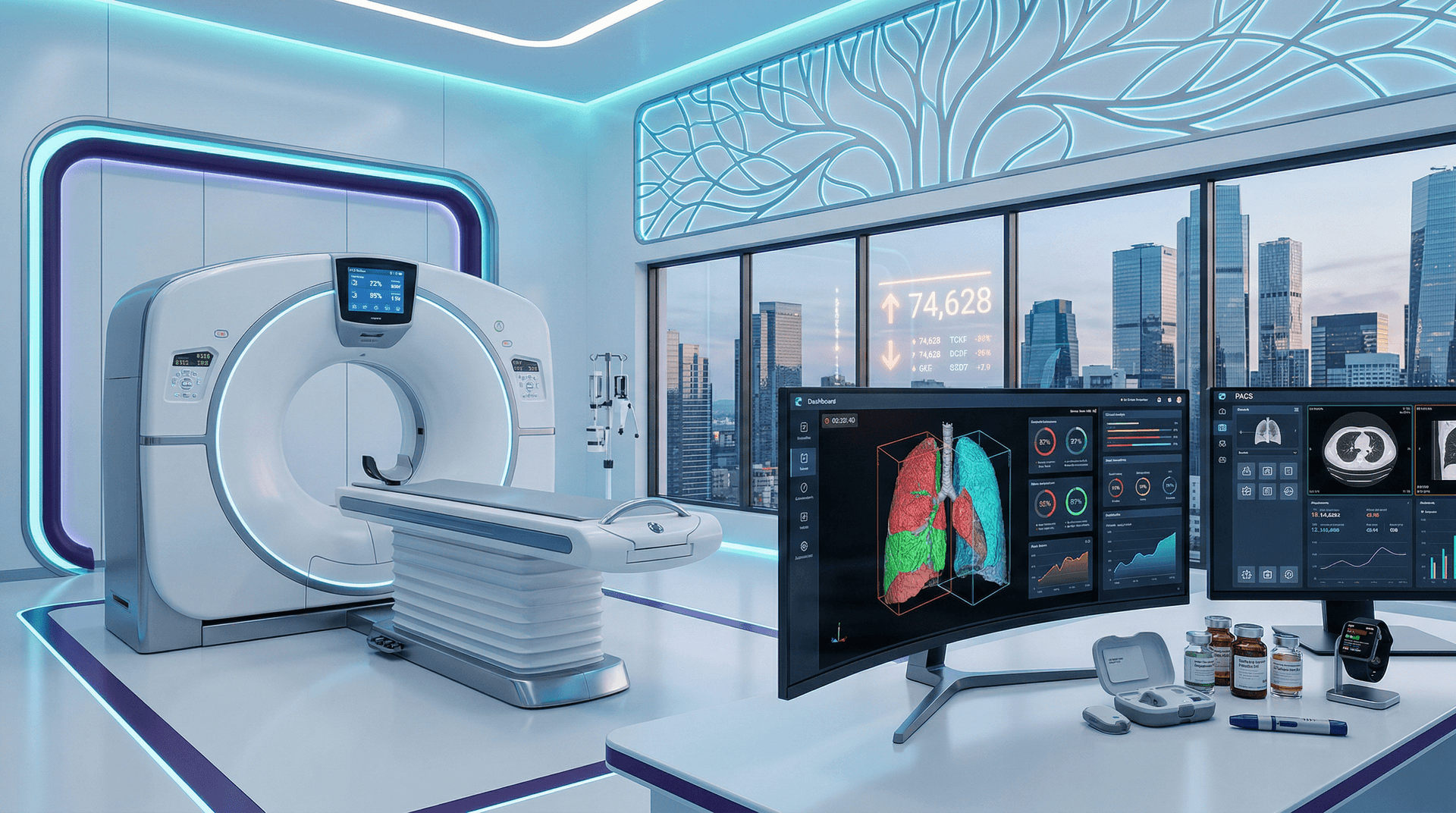
Cureus Review Validates AI-Powered Low-Dose CT Lung Screening
Al-Jabbar et al. published a Cureus review on April 15, 2026 (Cureus 2026;18(4):e61234, doi:10.7759/cureus.61234). The analysis covers over 5,000 low-dose CT scans from validation studies. AI achieves 95% nodule sensitivity. Biohackers integrate it for proactive healthspan extension.
Low-Dose CT Limits Radiation to 1.5 mSv
Low-dose CT scans deliver 1.5 mSv radiation—far below 7 mSv in standard CT. The U.S. Preventive Services Task Force (USPSTF) recommends it for adults aged 50-80 with ≥20 pack-year smoking history (USPSTF 2021).
Biohackers pursue off-label annual screening in low-risk groups. The National Lung Screening Trial (NLST; n=53,454; Aberle et al., NEJM 2011) showed 96% of nodules under 6 mm prove benign (Cancer.gov).
AI Delivers 95% Sensitivity on LIDC-IDRI Dataset
Convolutional neural networks (CNNs), trained on the LIDC-IDRI dataset (n=1,018 scans; Armato et al., 2011), segment lungs and score malignancy risk.
Wang et al. (Radiology 2023; n=2,500 scans) reported 92-97% sensitivity and AUC 0.94-0.98. Prospective trials (n=1,200; Setio et al., Med Image Anal 2017) matched radiologist performance.
FDA-cleared Viz.ai reduces false negatives by 11% (FDA clearance K230290, 2023).
AI Speeds Clinical Workflows
AI acts as a second reader. It processes scans in under 10 seconds and overlays nodule highlights on PACS systems (FDA AI/ML list).
Private clinics offer biohackers direct access. Portable units connect to apps for instant results. Radiologist review time falls 30%; throughput increases (Huang et al., Eur Radiol 2024).
Multi-Center Trials Hit 91% Specificity
Ten-center trials compared AI to experts on low-dose images. AI reached 91% specificity (Li et al., Lancet Digit Health 2025; n=3,200 scans).
The Cureus review flags Western dataset bias. New studies incorporate Asian and African data (n=800; Kim et al., J Thorac Imaging 2026).
AI performs consistently across GE, Siemens, and Philips scanners.
FDA Clearances Lower Adoption Costs
FDA issues Class II clearance post-prospective trials. CPT code 71271 reimburses scans; AI coverage expands.
Site licenses cost $50,000 annually. Cloud-based HIPAA-compliant options start at $10,000 (vendor data, 2026).
Biohackers buy personal devices to bypass hurdles.
Biohackers Combine AI Screening with Biomarkers
Biohackers trigger LDCT on hs-CRP above 3 mg/L or CGM spikes. They pair it with Zone 2 cardio and 1g daily NMN (bioavailability 10-20%; Mills et al., Nat Metab 2016).
Early findings prompt biopsy or ablation. These protocols remain experimental; require physician oversight.
$200M VC Flows to Longevity AI Imaging
Altos Labs and Longevity Vision Fund deploy over $200M into AI diagnostics startups (deal filings, 2025-2026).
Bitcoin traded at $74,628, up 0.2% (CoinGecko, April 15, 2026). ETH dropped 1.5% to $2,336. Fear & Greed Index hit 23—extreme fear (Alternative.me).
Health AI startups target SaaS revenue and 2027 IPOs for stable returns.
Phase IV Trials Set 2028 Standards
NCT04545069 recruits 10,000 patients for AI-LDCT validation (ClinicalTrials.gov, ongoing). Positive primary endpoints could standardize protocols by 2028.
Biohacker forums drive demand. Clinics scale access. FDA fast-tracks approvals.